MQF Level 5 CHE1700 Coursework
| CODE | CHE1700 |
| TITLE | Chemistry Practical 1 |
| UM LEVEL | 01 – Year 1 in Modular Undergraduate Course |
| MQF LEVEL | 5 |
| ECTS CREDITS | 4 |
| DEPARTMENT | Chemistry |
Experiment 10: Argentometric Methods
Introduction
Titration techniques play a crucial role in analytical chemistry, providing a quantitative means of determining the concentration of various ions in solution. The effective titration techniques to determine the concentration of metal cations are complexometric titrations. One such method of complexometric titrations is argentometric titration which is used to quantify the concentration of specific anions through their complexation with silver cations. Argentometric titrations are precipitation titrations as the silver salt which is one of the products of the titration reaction is a precipitate.
In this experiment three methods of argentometric titrations were used which are the Mohr method, Fajans method and Volhard method. The Volhard’s method is the most commonly used argentometric method.1 In this method a silver nitrate solution (standardised by two other argentometric methods) is titrated with a solution containing the thiocyanate ions.1 This method was the last one to be performed in this experiment as the aim was to determine the concentration of thiocyanate ions [SCN–] in a solution by this method. In this method iron(III) ammonium sulphate serves as an indicator.1 As shown in the following reversible ionic equation first a precipate of silver thiocyanate forms upon addition of thiocyanate solution.
Ag+(aq) + SCN−(aq) → AgSCN(aq)
With the first slight excess of thiocyanate ion a red colourisation of the solution occurs due to the formation of [FeSCN]2+ as the slight excess of silver reacts with the Fe3+ ions of the indicator. This is shown by the reversible ionic equation below.
Ag+(aq) + Fe3+(aq) → [FeSCN]2+(aq)
The very first method that was carried out was the Mohr method and following it was the Fajans method. These were the two methods used to standardise the silver nitrate solution. In the Mohr method, the small quantity of potassium chromate acts as the indicator for the argentometric titrations. The silver ions react with the chromate ions to form the brick red silver chromate (Ag2CrO4).
Chemicals Used
The chemicals provided during the experiment are listed with their respective details in Table 1 below:
| Chemical | Grade | Brand | Amount |
| Sodium chloride | Analar | Fischer | Grams or less |
| Silver nitrate | Analar | Aldrich | Millilitres or less |
| Potassium chromate | GPR | Hopkins and
Williams |
Millilitres or less |
| Fluorescein | GPR | Hopkins and
Williams |
Millilitres or less |
| Nitric acid | GPR | Fischer | Millilitres or less |
| Iron(III) ammonium sulfate indicator | GPR | Sigma | Millilitres or less |
| Potassium thiocyanate | – | – | Millilitres or less |
Table 1: Table containing details of the chemicals provided.
Apparatus and Materials Used
Stand and clamp, hotplate/stirrer, electronic balance, beakers, conical flasks, 250-mL volumetric flask, burette, 25-mL volumetric pipette, 50-mL volumetric pipette and weighing boat.
Methodology
The first method of the experiment was the Mohr Method for the standardisation of the approximately 0.02M silver nitrate solution provided. First about 0.3 g of oven-dried sodium chloride were weighed on a weighing boat using an electronic balance to record the mass reading. It was then transferred quantitatively to a 250-mL volumetric flask and was made up to the mark with distilled water. The stock solution was shaken for few seconds (around 10 times) to ensure a homogenous solution. 25 mL of the solution were than pipetted into a clean conical flask using a clean 25-mL volumetric pipette. 1 mL of 5% potassium chromate solution was then added to act as an indicator and the solution was titrated with silver nitrate solution whilst swirling the liquid. The endpoint was indicated by the appearance of the first reddishbrown colour in the solution. The titration was repeated until three concordant results were obtained.
The indicator blank was determined as follows: 1 mL of the indicator solution was added to 50 mL of distilled water into a clean conical flask. This indicator solution was titrated with a solution of silver nitrate that was obtained by diluting 10.0 mL of the 0.02M silver nitrate to 100 mL in a clean 100-mL volumetric flask. The endpoint had been reached when the colour of the blank was the same as that of the solution that had just been titrated. The blank titration was repeated until three concordant results were obtained. The mean blank titre had to be corrected to correspond to the value it employed. This adjusted value was subtracted from the mean volume of silver nitrate used in the titration. The molar concentration of silver nitrate was then calculated.
The second method was the Fluorescein Method for the standardisation of the approximately 0.02M silver nitrate solution provided. The procedure of the Mohr Method was repeated but instead, fluorescein was used as the indicator. 10 drops of it were added to the solution in a conical flask. The endpoint was indicated when the precipitated silver chloride assumed a pink colour. The titration was repeated until three concordant results were obtained.
The third method was the Volhard Method. First, 25 mL of the standardised silver nitrate solution was pipetted into a clean conical flask using a clean 25-mL volumetric pipette. 5 mL of 6M nitric acid and the ferric indicator (a saturated aqueous solution of iron(III) ammonium sulfate to which 2M nitric acid (10 drops) had been added) were added. This mixture was then titrated with the potassium thiocyanate solution provided. Near the endpoint, the silver thiocyanate was made flocculent and settled out. The endpoint was indicated by the appearance of a permanent faint brown colour. The titration was repeated until three concordant results were obtained.
Precautions
- Ensuring that there are no air bubbles.
- Consistent swirling during titration.
- If overshot the titration must be repeated.
Results
The titration results of this experiment are shown in the following tables.
Mass of sodium chloride: 0.308 g
| 1st titration | 2nd titration | 3rd titration | |
| Final burette reading (mL) | 26.45 | 26.45 | 26.50 |
| Initial burette reading (mL) | 0.00 | 0.00 | 0.00 |
| Titre value (mL) | 26.45 | 26.45 | 26.50 |
| Mean titre value: 26.47 mL of AgNO3 | |||
Table 2: Table containing the measurements taken during the experiment.
Indicator blank
| 1st titration | 2nd titration | 3rd titration | |
| Final burette reading (mL) | 41.00 | 41.05 | 41.05 |
| Initial burette reading (mL) | 38.05 | 38.05 | 38.05 |
| Titre value (mL) | 2.95 | 3.00 | 3.00 |
| Mean titre value: 2.98 mL of AgNO3 | |||
Table 3: Table containing the measurements taken during the experiment.
Fluorescein Method
| 1st titration | 2nd titration | 3rd titration | |
| Final burette reading (mL) | 26.30 | 28.20 | 27.90 |
| Initial burette reading (mL) | 0.40 | 2.30 | 2.00 |
| Titre value (mL) | 25.90 | 25.90 | 25.90 |
| Mean titre value: 25.90 mL of AgNO3 | |||
Table 4: Table containing the measurements taken during the experiment.
Volhard Method
| 1st titration | 2nd titration | 3rd titration | |
| Final burette reading (mL) | 27.60 | 27.60 | 30.80 |
| Initial burette reading (mL) | 2.00 | 2.00 | 5.20 |
| Titre value (mL) | 25.60 | 25.60 | 25.60 |
| Mean titre value: 25.60 mL of AgNO3 | |||
Table 5: Table containing the measurements taken during the experiment.
Observations
- Mohr methos: On titration of sodium chloride with the silver nitrate solution a peachcoloured endpopint was observed.
- Fajans methos: A white precipitate was formed and then turned pink.
- Volhard method: A permanent reddish-coloured endpoint was observed.
Discussion
The results from this experiment provide insight into he effectiveness and precision of argentometric titrations, particularly the Volhard method, in determining the concentration of thiocyanate ions ([SCN–]) in solution. The proper standardisation of the silver nitrate solution was very important as the accuracy of the titration was highly dependent on it. The standardisation of the silver nitrate solution as already mentioned in the introduction was performed using two different methods: the Mohr method and the Fajans. By comparing the values obtained from these standardization methods allowed for an evaluation of their precision and suitability in argentimetric titrations.
In this discussion, the experimental results will be analyzed in relation to the expected theoretical values, considering possible sources of error and limitations of the methods used. Additionally, the impact of experimental conditions—such as pH control, indicator effectiveness, and endpoint detection—on the overall accuracy of the titration will be examined. By evaluating these factors, we can assess the reliability of the Volhard titration and its practical applications in quantitative analysis.
As mentioned in the introduction in the Fajans method an adsorption indicator is used. It is a substance that adsorbs onto or desorbs on from the surface of the substance in a precipiation titration. In this experiment the Fajans method the adsorption indicator that was used is fluoresecin. Its molecular structure is shown in Figure 1.
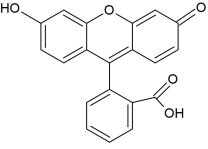
Figure 1: Figure showing the molecular formula of fluorescein
The term adsorption is when the particles of the substance sticks to the surface of the fluorescein molecule
Need Expert Help
If you are struggling to complete your CHE1700 Chemistry Practical 1 Coursework (University of Malta), especially with titration calculations, data analysis, or writing a clear lab report discussion, you can choose Malta Assignment Help like many other students. Our experts provide chemistry assignment help tailored to your coursework requirements with accurate and well-structured solutions. You can also review our mqf level 5 assignment example to check the quality of our work. Order our cheap assignment help today and receive a 100% custom, human-written, and plagiarism-free solution.
